POSTER ABSTRACTS
Materials should NOT be shared with those that are not registered for the conference. Poster abstracts are not proofed for spelling and/or grammar errors.
The poster and/or other information contained on this website may NOT be downloaded and/or used without prior written permission from all authors on the project. If you would like to be connected with the author(s), please email [email protected].
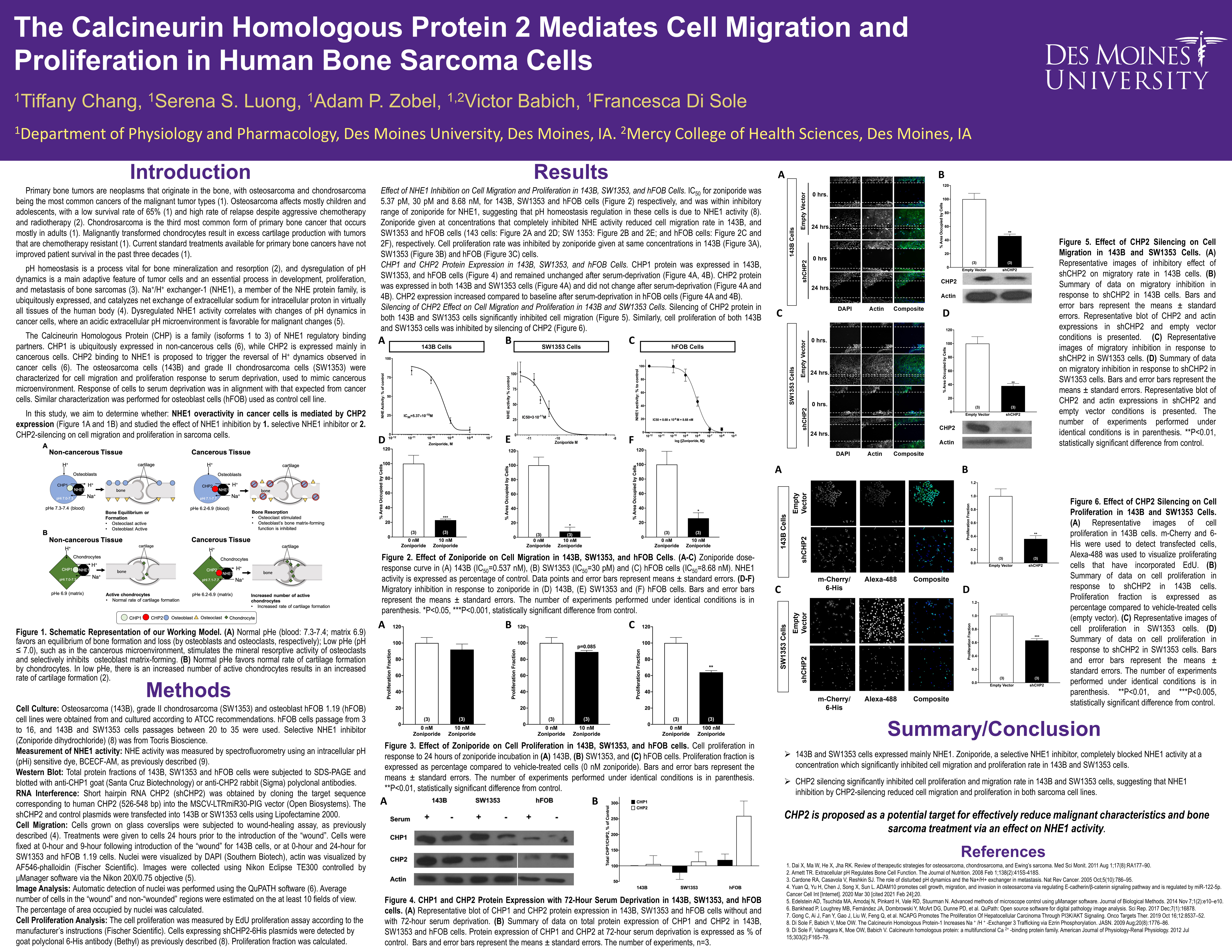
The Calcineurin Homologous Protein 2 Mediates Cell Migration and Proliferation in Human Bone Sarcoma Cells
1Tiffany Chang, MSBS, DO‘23, 1Serena S. Luong, DO, 1Adam P. Zobel, DO,1,2Victor Babich, PhD, and 1Francesca Di Sole, PhD
1Physiology and Pharmacology Department, Des Moines University, Des Moines, IA.
2Mercy College of Health Sciences, Des Moines, Iowa
Abstract
Bone sarcomas are malignant tumors including osteosarcoma and chondrosarcoma and their current treatments have not improved patients’ survival. Extracellular acidosis inhibits osteoblasts, weakening bone matrix, and stimulates chondrocytes, expanding cartilage matrix and may contribute to the sarcoma pathogenesis. Extracellular acidosis in cancer due to dysregulation of pH dynamics is mediated by the Na+/H+ exchanger-1 (NHE1) overactivity, the main transporter responsible for pH regulation, and promotes malignant cellular migration and proliferation. We hypothesize that NHE1 overactivity in cancer cells is mediated by the Calcineurin Homologous Protein 2 (CHP2), regulator of NHE1 function, and studied NHE1 inhibition by CHP2-silencing on migration and proliferation in sarcoma cells. We demonstrated that: 1. decrease in cell migration and proliferation by serum deprivation, used to mimic cancerous microenvironment, was only transient in osteosarcoma (143B), and chondrosarcoma (SW1353) cells in alignment with malignant characteristics expected in cancer cells. Indeed, cell proliferation was inhibited by serum deprivation in osteoblast (hFOB) cells used as control. 2. 143B and SW1353 cells expressed mainly NHE1. Zoniporide, a selective NHE1 inhibitor, completely blocked NHE1 activity at a concentration which significantly inhibited migration and proliferation in 143B and SW1353 cells, respectively 3. CHP2 protein expression was induced by serum deprivation in hFOB cells and silencing of CHP2 significantly inhibited cell proliferation and migration in both sarcoma cell lines. Our data suggest that NHE1 inhibition by CHP2-silencing reduced cell migration and proliferation in 143B and SW1353 cells. CHP2 is proposed as a potential target for effectively reduce malignant characteristics and for bone sarcoma treatment.
Key Words: bone sarcoma, cancer, migration, proliferation, Na/H Exchanger, Calcineurin Homologous Protein
Support: Iowa Osteopathic Education and Research Funds
Access PDF version to expand view.